Collagen denaturation is initiated upon tissue yield in both positional and energy-storing tendons
A great new article from a collaboration between Dr. Jeff Weiss's lab and our founder, Dr. Michael Yu's lab at the University of Utah. The new Acta Biomaterialia paper explores at what point collagen denaturation occurs in tendons under mechanical strain and how tendon type influences the mechanism of failure. Researchers evaluated functionally distinct tendons using positional tendons (rat tail tendons) and energy-storing tendons (rat flexor digitorum longus tendons) and saw that both tendons had permanent collagen denaturation begin at the yield strain and both had the same mechanism of failure. Surprisingly, they showed that although the mechanism was the same, the amount of denatured collagen found in energy-storing tendons was half the amount found in positional tendons at tissue failure. This can be explained by the different enzymatic collagen crosslink profiles between the two types of tendons. Understanding the onset and accumulation of denatured collagen in tendons relative to applied stretch is essential to understanding tendon failure
ABSTRACT
Tendons are collagenous soft tissues that transmit loads between muscles and bones. Depending on their anatomical function, tendons are classified as positional or energy-storing with differing biomechanical and biochemical properties. We recently demonstrated that during monotonic stretch of positional tendons, permanent denatured collagen begins accumulating upon departing the linear region of the stress-strain curve.
However, it is unknown if this observation is true during mechanical overload of other types of tendons. Therefore, the purpose of this study was to investigate the onset of collagen denaturation relative to applied strain, and whether it differs between the two tendon types. Rat tail tendon (RTT) fascicles and rat flexor digitorum longus (FDL) tendons represented positional and energy-storing tendons, respectively.
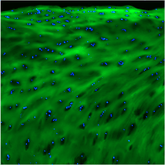
The samples were stretched to incremental levels of strain, then stained with fluorescently labeled collagen hybridizing peptides (CHPs); the CHP fluorescence was measured to quantify denatured collagen. Denatured collagen in both positional and energy-storing tendons began to increase at the yield strain, upon leaving the linear region of the stress-strain curve as the sample started to permanently deform.
Despite significant differences between the two tendon types, it appears that collagen denaturation is initiated at tissue yield during monotonic stretch, and the fundamental mechanism of failure is the same for the two types of tendons. At tissue failure, positional tendons had double the percentage of denatured collagen compared to energy-storing tendons, with no difference between 0% control groups. These results help to elucidate the etiology of subfailure injury and rupture in functionally distinct tendons.
CHECK OUT THE FULL ARTICLE HERE