Using CHP to Understand Cardiac Fibrosis Pathology and Therapeutic Efficacy
Patients that survive an acute Myocardial Infarction (MI) are at a higher risk for subsequent cardiac fibrosis. Directly following a heart attack, myofibroblasts deposit significant amounts of collagen - which is vital for establishing an initial scar and maintaining the mechanical integrity of the heart. However, this process often goes too far, depositing excessive collagen (fibrosis) that leads to eventual heart failure. This indicates a therapeutic need to specifically reduce cardiac fibrosis following MI.
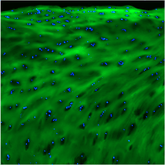
Recent research identified a particular cell surface protein - ANTXR1 - that is upregulated following MI and stimulates excessive collagen deposition. Collagen Hybridizing Peptides (CHPs) bind to remodeling and damaged collagen, making them the perfect tool to visualize sites of excessive collagen deposition in a post-MI patient heart tissue. Costaining ANTXR1 and damaged collagen with CHP was used to assess whether ANTXR1 colocalizes with cardiac fibrosis. In the figure below, TNNI3 (red, troponin1) stains healthy, cardiomyocyte-rich regions, where ANTXR1 (green) and CHP (yellow) staining is indicative of scar tissue. These results confirmed that the biomarker ANTXR1 is highly colocalized with fibrotic collagen, validating it as a therapeutic target for cardiac fibrosis.

Next, the authors used CHPs to evaluate the efficacy of a therapeutic blockade of ANTXR1 using antagonistic antibodies (T8Ab) against ANTXR1 in murine models. In short, MI-induced mice were treated with the antagonist antibodies for 6 weeks, and various readouts for cardiac fibrosis were taken to assess efficacy. It was found the antibody-treated mice had better survival and better heart function, and CHP staining indicated lower fibrotic collagen deposition. The figure below shows CHP staining (red) rising dramatically in MI-induced mice, but this staining is lower with therapeutic antibody treatment.

CHPs were integral to validate ANTXR1 as a therapeutic target and to assess the therapeutic efficacy of antibody antagonists. The ability to specifically bind to damaged collagen in heart tissues provided valuable insight on the molecular state of this collagen, granting deep understanding of cardiac fibrosis pathology and advancing therapeutic development efforts.
Full Paper: Boccella, Nicola, et al. "ANTXR1 blockade enhances cardiac function in preclinical models of heart failure." Nature Cardiovascular Research (2025): 1-18.