Wound healing breakthrough: How Collagen Hybridizing Peptides turn damaged tissue into a target for drug delivery.
Wound infections pose a major risk to recovery, having the potential to turn otherwise minor injuries into severe medical challenges. Scientists have continued to investigate the potential of Antimicrobial Peptides (AMPs) for their ability to kill a broad spectrum of microbes while mitigating the risk of increasing microbe resistance.
While powerful, AMPs do have critical flaws. When applied to wound areas, AMPs are quickly washed away, concerning due to the harmful effects AMPs can have on healthy tissue. AMPs have great therapeutic potential, but they are held back by a lack of targeted delivery and retention. But what if we could anchor AMPs directly to the injury site to create a localized, sustained antimicrobial defense? Researchers demonstrate that, by conjugating an AMP to our Collagen Hybridizing Peptides, we can selectively target and bind AMPs to sites of tissue damage for targeted, sustained antimicrobial activity.

The first step researchers took was to directly link 1 AMP to 1 CHP, but this decreased the molecule’s antimicrobial activity. Only when the researchers utilized a dimeric structure, conjugating 2 AMPs for every 1 CHP, did researchers see success. Not only did this new dimeric peptide exhibit AMP’s antimicrobial properties; it was more effective at killing a broad spectrum of microbes, including S. Aureus and P. Aeruginosa, than the original AMP alone.
Continue Reading
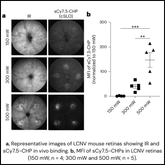
Next, the researchers sought to confirm CHPs’ targeting ability, and, as expected, the CHP-AMP conjugate selectively bound to damaged collagen, enabling a longer, targeted AMP effect. Critically, CHPs showed no binding to healthy, intact tissue, confirming CHPs act as precise guides taking the AMP directly to the injury site.
With a successful design in hand, researchers then tested the conjugate in a preclinical rat wound model with remarkable results. Researchers noted:
Sustained Antimicrobial Activity: Wounds treated with a single dose of CHP-AMP conjugates showed significantly lower bacterial growth 24 hours post-treatment when compared to wounds treated with unconjugated AMP. CHPs effectively created a localized reservoir of AMPs to continuously fight infection.
Accelerated Healing: By providing a stable antimicrobial defense, the CHP-AMP conjugate drastically improved healing outcomes. Wounds treated with CHP-AMP conjugates closed faster, exhibited less inflammation, and showed more complete tissue regeneration. Furthermore, CHP-AMP-treated wounds began forming new hair follicles, a key sign of high-quality wound healing.

Proven Safety: Throughout the study, CHPs showed no signs of local or systemic toxicity, underscoring the benefit of keeping the therapeutic localized to its injury site and away from healthy tissue.
Throughout the study, CHPs showed no signs of local or systemic toxicity, underscoring the benefit of keeping the therapeutic localized to its injury site and away from healthy tissue.
The implications of this breakthrough extend far beyond wound care. This strategy provides a blueprint for utilizing CHPs as a targeted drug delivery platform. Any disease characterized by collagen damage, from fibrosis to arthritis and beyond, could potentially be treated with a CHP-guided approach, opening a new frontier in targeted drug delivery.
Mo, Xiaoyun, et al. "Targeting collagen damage for sustained in situ antimicrobial activities." Journal of Controlled Release 360 (2023): 122-132.