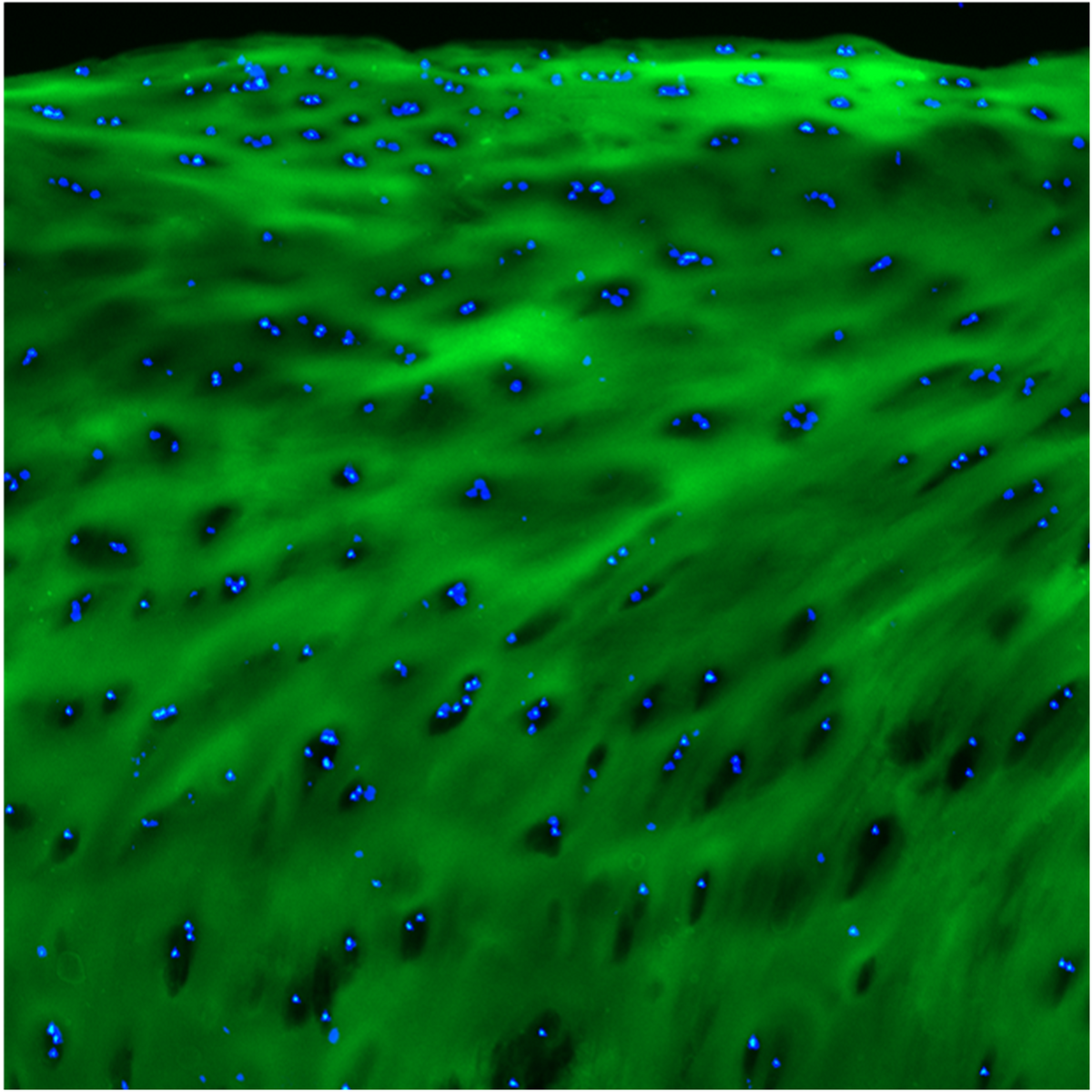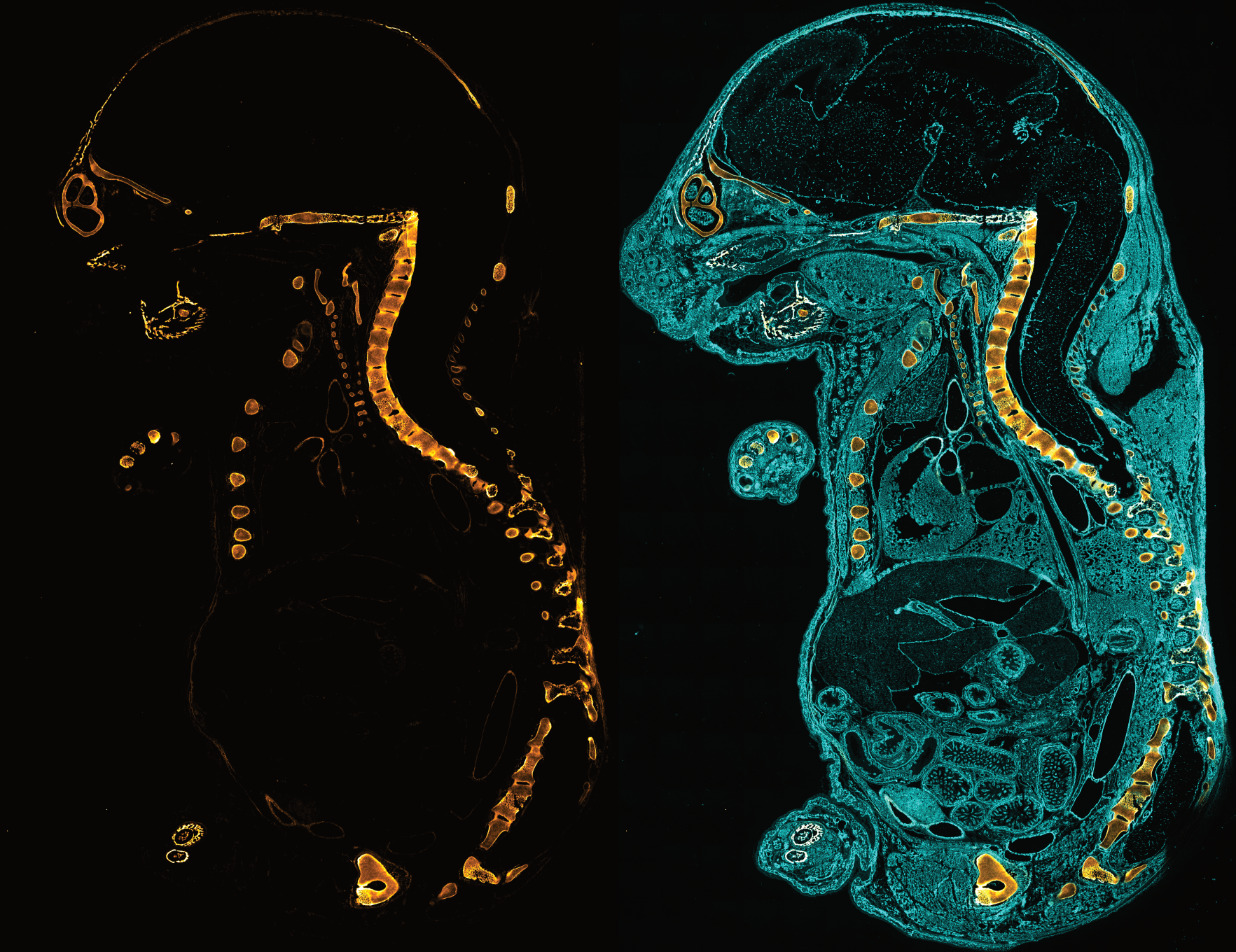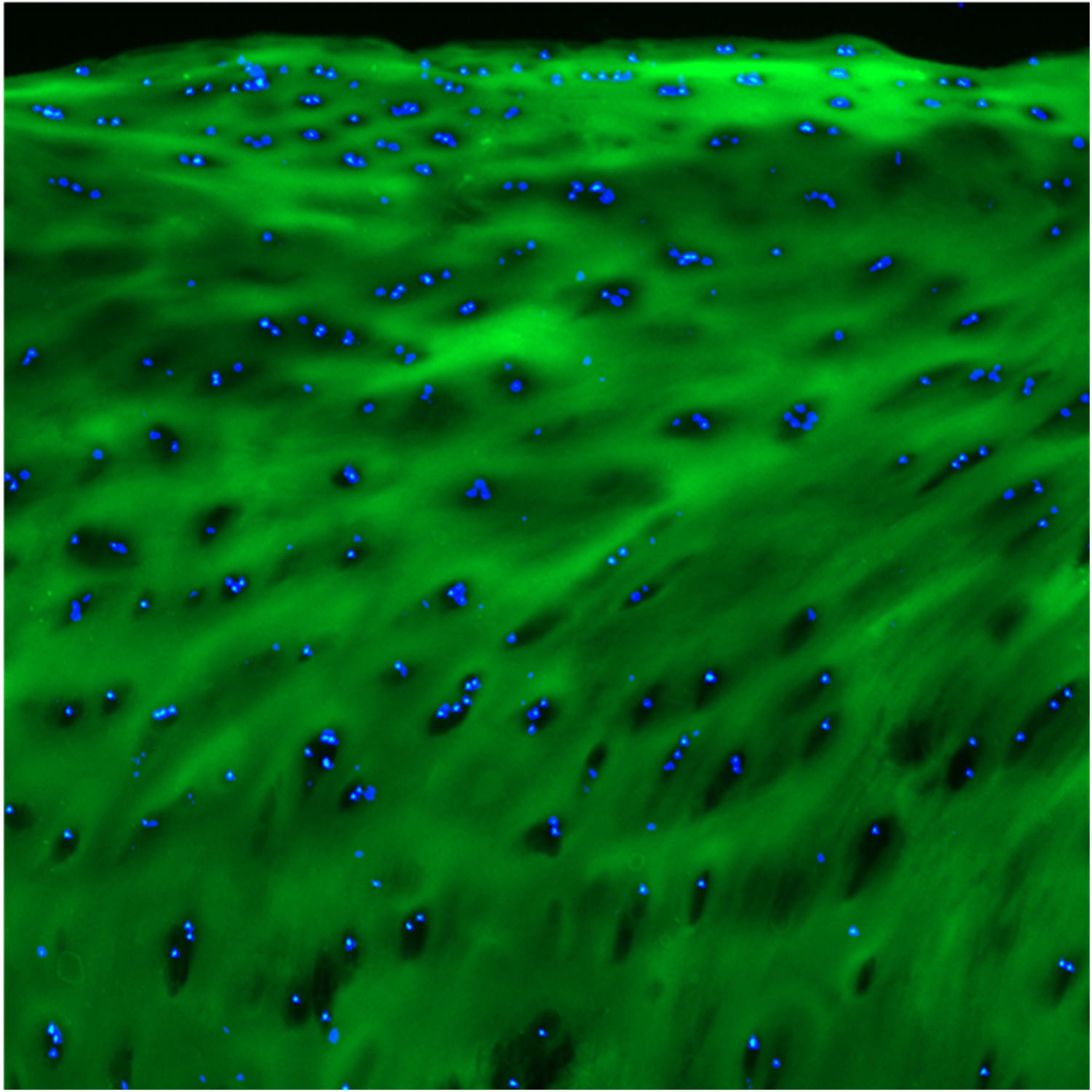
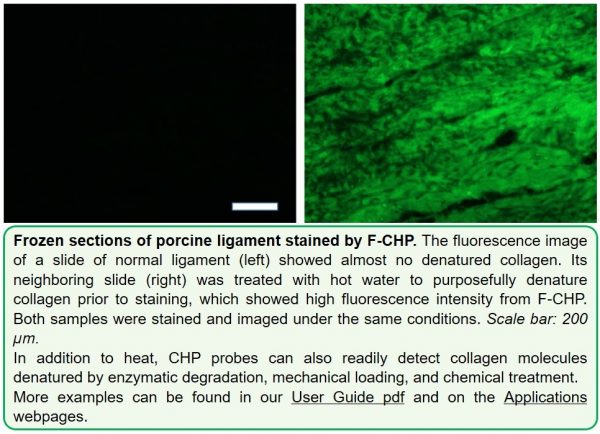
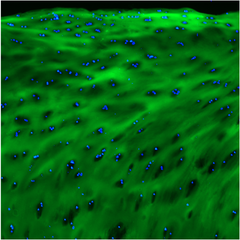
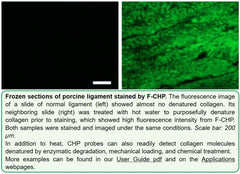

F-CHP | Collagen Hybridizing Peptide, 5-FAM Conjugate
- $218.00 USD
- $218.00 USD
- Unit price
- per
Subtotal:
$218.00
Couldn't load pickup availability
Description
xDescription
User Guide with FAQ
xShipping & Returns
xORDER PROCESSING
Please allow 1-2 Business Days to process your order. Expect an email within 2 Business Days to let you know your order is on the way!
LOCAL PICKUP - 675 S Arapeen Drive, Salt Lake City, UT 84108
Orders selected for local pickup can be picked up Monday- Friday, from 9am-4pm. An email will be sent when your order is ready for pickup.
FLAT RATE SHIPPING DOMESTIC & INTERNATIONAL
Orders are generally shipped within 1-2 Business Days using FedEx 2 Day or FedEx International Priority. Flat rate shipping fee of $39 applies to orders shipping to USA. Flat rate shipping fee of $77 applies to orders shipping WORDLWIDE. You may also opt to ‘Use Your Own FedEx Account’ at checkout and shipping will be billed directly to your account.
REFUNDS, RETURNS, AND EXCHANGES
We do not accept returns of any kind.
In the event that your order arrives damaged in any way, please email us as soon as possible at orders@3helix.com with your order number and a photo of the item’s condition. We address these on a case-by-case basis but will try our best to work towards a satisfactory solution.
If you have any further questions, please don't hesitate to contact us at orders@3helix.com.
Related Products
3Helix
3Helix
3Helix
3Helix
3Helix
3Helix
3Helix
3Helix
3Helix
3Helix
Recently Viewed Products

- Choosing a selection results in a full page refresh.